It’s day 6 of National Breast Cancer Awareness Month. I’m creating a new post every day in the Month of October to spread awareness, information, accessible science, practical advice, and humor. I can write about the science of breast cancer all day, and I’ve got plenty of stories to tell and woo woo bullshit to debunk, but I really want to hear from y’all.

What do you want to know about breast cancer, breast health, breast biology, treatments, surgeries, radiation, or any topic? Want me to investigate something a friend told you would help with your cancer, or some off the wall thing you found on FaceBook? Need resources on getting screening mammograms, including financial assistance, and on getting breast healthcare? Want to see pictures of my cats and hear about how they’ve kept me entertained and happy during my breast cancer years?
Let me know!
I’m a research whore – send me down a rabbit hole and help me find something new! Comment on this post with what you want to know and I’ll select three folks at random to receive a signed copy of Talking to My Tatas, my breast cancer book.

For today’s topic, let’s talk about the DESTINY-4 clinical trial that represents a HUGE advance in breast cancer treatment options for people with HER2-low advanced cancer. As I discussed in my previous post about breast cancer subtypes, HER2+ breast cancers are diagnosed based on how they look under the microscope and if they have extra copies of the gene that makes the HER2 cell surface receptor protein and/or if they express higher than normal levels of HER2, a protein that makes breast cancer cells grow uncontrollably.
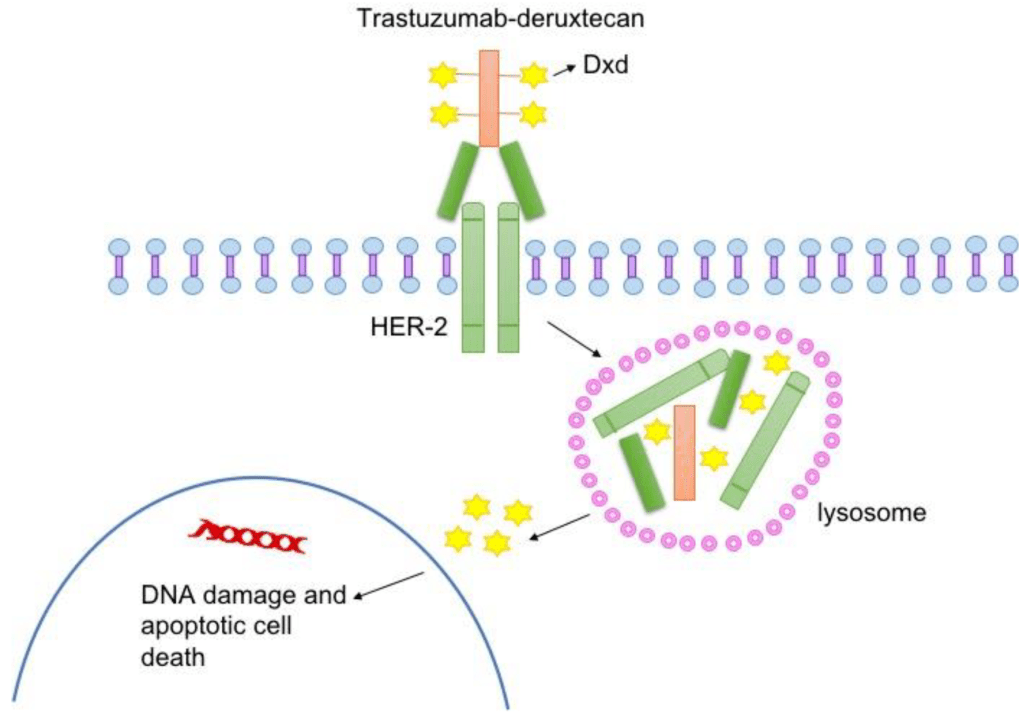
The standard of care is to classify HER2+ breast cancers as meeting a threshold for HER2 receptor expression. Cancers that express levels of HER2 below that threshold were considered HER2-negative and not treated with HER2 targeted therapies. But people with low levels of HER2 in their breast tumor could still benefit from HER2-targeted therapies. To test that hypothesis, Dr. Shanu Modi, MD, of the Memorial Sloan Kettering Cancer Center led a clinical trial that tested an antibody drug conjugate called Trastuzumab Deruxtecan (T-DXd) to see if patients with HER2-low metastatic breast cancer benefitted from this treatment. This drug consists of trastuzumab, an antibody that binds to HER2 on tumor cells, and a toxic drug that is delivered directly to the tumor by trastuzumab, which kills cancer cells. People who received this drug in the trial lived longer and had a longer time before their cancer progressed compared to patients who received chemotherapy. The drug reduced tumor burden, the amount of cancer in the body. The major side effects of treatment with this drug include heart and lung function issues, which need to be closely monitored in people taking this drug.
Not only do these results provide hope and more treatment options for patients with metastatic breast cancer, they will likely change the way we classify breast cancers by molecular subtype, adding HER2-low to the classification system. Ongoing clinical trials will test this drug on patients with different levels of HER2 expression to determine the range of expression that defines HER2-low tumors likely to respond.
To learn more about Trastuzumab Deruxtecan, visit the National Cancer Institute website. Click here for more information about breast cancer treatment clinical trials.