If I had a quarter for every time someone told me they read that some discovery was the cure for cancer over the past twenty years, I could afford to take that trip to Tahiti I’ve been dreaming about. Science and the news media have an interesting relationship. On one hand, it’s always great to get coverage for advances in science. It keeps the public informed and engaged, which in turn means more interest and research dollars for laboratory and clinical investigation.
On the other hand, the news media gets a lot wrong, and that’s actually bad for keeping the public informed and for public perception and expectations, especially when it comes to complex diseases like cancer.
Take, for example, recent press coverage of a discovery related to tumor immunology – the study of how we can harness a patient’s own immune system to fight their cancer. It’s a hot topic. In fact, two leaders in the field, James Allison and Tasuku Honjo, were awarded the Nobel Prize for Physiology and Medicine in 2018 for their discovery of a new cancer therapy by inhibition of negative immune regulation, which led to the development of several drugs currently available to treat certain types of cancer. More recently, a paper published in Nature Immunology captured the attention of news outlets, leading to headlines like:

The first outlet, Science Alert, gets it right. The discovery is indeed remarkable, and the note about the ability to kill several cancer types in the laboratory is an accurate representation of what the study showed (my only issue with the headline is the clunky construction – discovery doesn’t kill anything – but that’s just me being nitpicky). The study tested activity of T-cells, part of the immune system that kills cells that have been infected by pathogens (bacteria and viruses) that can also be engineered to target cancer cells in cell culture (cells grown on a plastic dish in the laboratory) and in mouse models (mice engineered to make tumors or transplanted with tumors). This is an essential first step for the development of new therapies, but it is a far cry from being ready to use in patients, as the BBC News and Newsweek headlines might lead people to believe.
But can switching to these new T-cells save me 15% or more on car insurance?
I get why this happens. In the age of 24 hour news cycles, multiple media outlets (online, television, radio, and print), sensational headlines sell. The idea that a new discovery could treat all cancer (a far-fetched notion given that cancer is a collection of diseases that are unique and adaptable) sounds exciting. It captures public interest, especially in patient and survivor communities. But those headlines are misleading, and that’s a problem. While most of the public will forget the specifics, some will look at the next headline related to cancer and think, “Wait, didn’t they find something that’s going to cure cancer soon? What happened to that?” The false promises made by these headlines can give the public the idea that most of what cancer researchers are doing is a waste since the sensationalized discoveries didn’t live up to the hype.
So, let’s look beyond the headlines and delve into the study. What did the investigators do (experimental methods and models), what were the results (data), and what do the results tells us (interpretation)?
To begin, we need a basic understanding of how T-cells (one of many cell types in the immune system) function in fighting disease. They are part of the adaptive immune response – meaning they are selected to attack specific pathogens based on unique proteins and, as noted in the paper, metabolic by-products, produced by target cells.
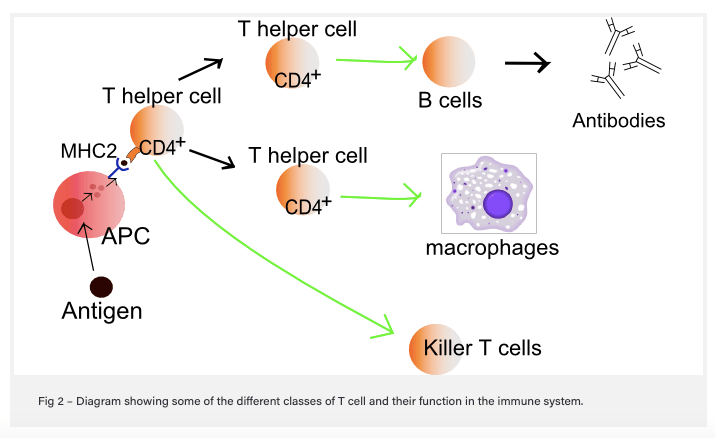
Here’s how it works: when a cell becomes infected with a bacteria or virus, the infected cell takes some of the foreign proteins (antigens) from the bacteria or virus and displays it on the cell surface in combination with proteins (major histocompatibility complex [MHC] – also known as the human leukocyte antigen [HLA] system in humans] that communicate with immune cells. Surveillance cells like macrophages, which can “eat” infected cells, also do this, and these cells are known as professional antigen-presenting cells (APC). Helper T-cells in the vicinity that recognize the antigen communicate with B-cells, which make antibodies to attack cells that are infected and display the same antigen, and with Killer T-cells (cytotoxic), which bind to and destroy infected cells that express the same antigen. In order to bind to the antigen/MHC complex, T-cells like the killers use cell surface protein receptors (T-cell receptors [TCR]). This is how conventional T-cells function.
Now, on to breaking down the actual published study!
The new study focused on unconventional T-cells that do not recognize protein antigens bound to MHC on antigen-presenting cells. These specialized T-cells normally sense bacterial metabolic by-products bound to the evolutionarily conserved, monomorphic MHC class 1-related protein MR1 – a fancy way of saying that the MR1 cell surface receptor is similar across species (evolutionarily conserved) and come in one form (monomorphic). This types of T-cell was identified from an experimental screen used to identify tumor-educated (e.g. grown in response to tumor cells) T-cells that recognize and kill cancer cells in a petri dish in a non-MHC (MHC mismatched) manner. This is important, since MHC/HLA are some of the most variable proteins within and between people and therefore not easy to develop and exploit as a universal anti-cancer treatment. The T-cell clone identified, called MC.7.G5, was able to kill cells from different types of cancer (lung, melanoma, leukemia, colon, breast, prostate, bone and ovarian) with different MHC/HLA types in petri dishes without harming normal, healthy cells.
Using other molecular biology techniques, the investigators determined that the T-cells that can recognize and destroy cancer cells in a non-MHC-dependent manner. Rather, the cancer-killing T-cells had receptors (TCR) that bound to MR1 plus a cancer-specific cargo (antigen – possibly a non-protein antigen) that has not yet been identified.
To see if these T-cells kill cancer in a whole animal model system, the investigators transplanted human leukemia cells into mice that lacked a functional immune system along with MC.7.G5 T-cells. The mice that received the MC.7.G5 T-cells showed evidence of reduced leukemia cells relative to controls, and when they tested leukemia cells that did not express MR1, the anti-leukemia activity went away – this is an important control, because it shows that MR1 is, in fact, necessary for MC.7.G5 T-cell activity. Finally, the investigators purified T-cells from actual patients with stage IV (metastatic) melanoma, engineered the purified T-cells to express the same T-cell receptor as MC.7.G5 cells, and tested the ability of these engineered T-cells to kill melanoma cells in a petri dish. The cells engineered to express the MC.7.G5 TCR killed melanoma cells from patients with similar or dissimilar HLA, but not melanoma cells that didn’t express MR1.
So what does all of this mean?? The significance is that T-cell subtypes that may be present in most humans and similar in most humans can recognize and destroy a variety of different cancer cells in laboratory models. It is exciting because, if validated in more laboratory research models (and if these types of T-cells are observed in actual cancer patients, including those with better outcomes), it could give scientists and physicians a new therapeutic tool to use in the clinic.
BUT
Before that can happen, a LOT more work has to be done, including:
Figuring out what cancer-specific antigen MR1 binds to – this is super important, since the presence of whatever that antigen might be would have to be detected in actual cancer patients before trying out these new T-cells in clinical trials.
Long-term studies to see if the health of the animal is affected by exposure to T-cell therapy. One of the challenges with anti-tumor immune therapy is the risk that patients might develop an auto-immune disease (e.g. immune cells will recognize the patient’s own healthy cells and attack, like what happens in Type I diabetes and Rheumatoid Arthritis). Remember, cancer cells are normal cells gone rogue, and they may be similar enough to normal cells within the body to trigger an unwanted immune response.
Getting the T-cells to go to the tumor, infiltrate it, and kill enough tumor cells is also a challenge. It’s a challenge with all immune therapies. Tumors that normally have a lot of T-cells hanging around (“hot” tumors) generally respond better to existing immune activating drugs, but many tumors don’t have a lot of T-cells that infiltrate (“cold” tumors”). These new T-cells can only be effective if they can get to the tumor, and tumors often adapt to evade or repel immune cells.
Finally, just because something works in laboratory models, like cells in petri dishes or even laboratory mice, doesn’t mean it will work in humans. We’ve been successfully killing cancer in petri dishes and mice for decades, and the clinical trials graveyard is full of therapies that showed great promise in the laboratory only to fail in clinical trials – some of those cases may be due to trial design, especially in early trials where patients weren’t always screened for the target. But other cases are probably due to our inability to replicate human disease fully in models that are not as naturally complex. Most mouse models used in the laboratory are inbred (for more on the history behind that, follow this link – yes, I know it’s a Wikipedia link. Wiki isn’t always wrong.). This means we’re using clones in our experiments, and some of those are even more abnormal since they don’t have a functional immune system. The advantage is that we can get more consistent, reproducible results and also that we can get mice without an immune system to grow human tumor cells without rejecting them. The disadvantage is that humans, not being clones, vary widely in their physiologies and responses to therapy. And even when it comes to mouse tumors grown in mice with a working immune system, there are differences in how mouse and human immune systems work – actually, in my experience, laboratory mice are WAY more resilient and resistant to infections than people.
Take home message – look past the headline to the actual data.
It’s a lot of work and requires a fair amount of background knowledge to go to the primary literature and make sense of it, but often enough, the story below the headline can give you enough information to figure out what a study shows, what it doesn’t show, and how close the results are to making it to the clinic.
For example, the BBC News article states that “The findings, published in Nature Immunology, have not been tested in patients, but the researchers say they have “enormous potential”.” I’ll also give the author of this article props for explaining CAR-T cell technology – which is how the cancer-fighting T-cells may be engineered from patient T-cells if the study findings are translated to the clinic. But the headline? Totally misleading.
Same goes for Newsweek, which presents the full headline as: ‘ONE-SIZE-FITS-ALL’ CANCER TREATMENT COULD BE ON THE HORIZON AFTER SCIENTISTS DISCOVER NEW IMMUNE CELL BY ACCIDENT.” Not the accident part – scientists discover stuff by accident all the time, but the idea that this is “on the horizon” gives the impression that it’ll be going to the clinic very soon. Um…not likely. Still, within the article, the author notes, “In mice experiments and in lab dishes, the team showed that a new type of what are known as T-cells could detect a range of cancerous cells, while differentiating them from healthy cells.” That, in a nutshell, is what the investigators actually showed. Headline is still misleading, though.
Bottom line – look BEYOND the sensational headline. It’s purpose is to hook you and get you to click on and read the article (while being bombarded with ads). Dig into the actual article to find out what the new and exciting study shows.
Pingback: Metastasis 101: How Breast Cancers Spread – Talking Tatas