This year I have the great fortune of mentoring a talented and dedicated medical student in my lab, Kalin Wilson. Her interest is in oncology, so it’s a great fit for my ongoing and new research directions. She’s working on two projects with similar goals: to identify and characterize new drug combinations and new experimental therapeutics for triple negative breast cancer in pre-clinical studies. This is an urgent unmet need in the clinic. Triple negative disease is a subtype of breast cancers that do not express hormone receptors (estrogen receptor and progesterone receptor) or cell surface HER2 (amplified in ~25% of breast cancers). These receptors are druggable targets, and their absence limits treatment options for patients with triple negative breast cancer to surgery, radiation, and chemotherapy. Triple negative breast cancers are aggressive and disproportionately affect young women and women of African descent. Our goal is to find molecular targets for new drugs to give women with this type of breast cancer more and better options.
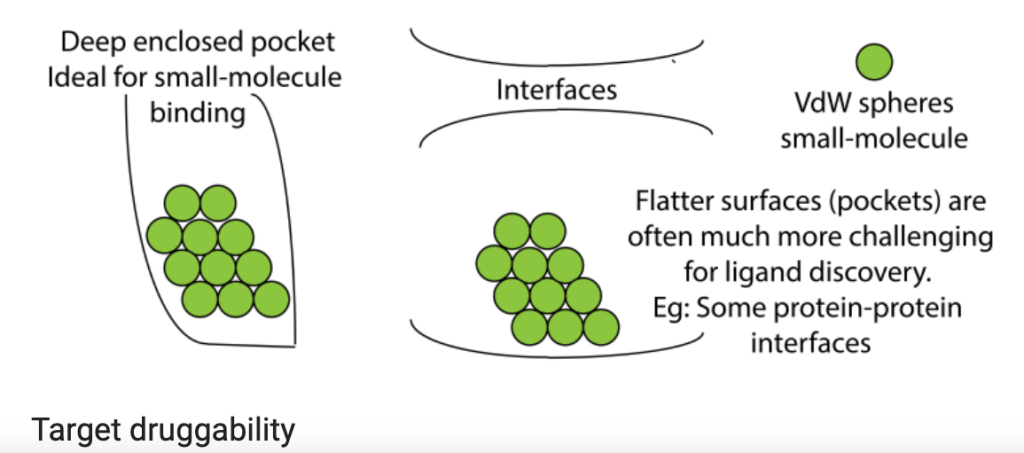
My student’s primary project is to test nanoparticle delivery systems to transport gene therapy to tumors. Many of the genes that drive cancer code for proteins that aren’t easily druggable by small molecules that fit neatly into a unique structural region in the target protein to block its function (e.g. deep enclosed pocket versus flat, relatively uniform interface or surface, as shown in the figure above). But what if we could stop production of cancer-driving proteins at the level of gene expression? This is actually possible in the laboratory setting in a process that exploits messenger RNA, the protein-making instructions that are copied from DNA and used by protein producing cellular machinery (see figure below). The use of small interfering RNA (siRNA) gene therapy, which causes the messenger RNA that encodes the protein’s blueprint to be destroyed, can theoretically stop production of any protein, which would make any target druggable. One of the challenges, however, is delivery of siRNAs to tumors. siRNAs tend to be unstable, so they can be easily destroyed by immune cells or taken up by the liver or kidneys as a part of their normal clearance functions. To overcome those delivery barriers, many biomedical engineers are applying nanotechnology, designing nanoparticles that surround the siRNA molecules. These nanoparticles shield and protect the siRNAs in circulation and can be modified to help homing to the tumor. In collaboration with Dr. Craig Duvall, we are testing nanoparticles delivering siRNA to destroy the blueprint for Rictor, a protein that we believe is essential for tumor cells to grow and prevents them from dying when they’re supposed to. Results so far are promising!
What I hope to give Kalin is a research experience that feeds her passion for science and drug discovery, to foster her natural skills and curiosity, and to keep striving for the goal of bench-to-bedside translational research. What she’s given me is her clinical perspective, something that has enriched my research and inspired me to do more directly translational research with the goal of clinical application. She’s also given me the gift of fearlessness and enthusiasm, which young scientists always possess in abundance and, fortunately, is contagious. The rewards of mentoring the next generation of scientists are many, but the synergy between experience (mentor) and fresh ideas and perspectives (mentee) is perhaps the most valuable.